Chemical Energy is energy stored in substances. It can also be considered as the potential energy. Chemical energy can in fact be calculated by adding up the bond energies present in a molecule.
What is a bond energy?
A bond energy is the minimum energy required to break one mole of bonds between two atoms.
A bond energy is measured in kilojoules.
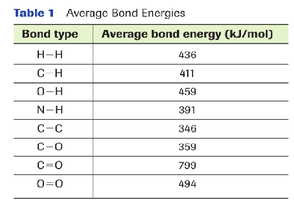
The table above lists some bond energies of various chemical bonds.
This table can be used to calculate different potential energies that are present in molecules. For example, let us calculate the potential energy present in 1 molecule of water.
1. One molecule of water has two O-H bonds.
2. Look at the table to determine the average bond energy of an O-H bond.
3. The average bond energy of an O-H bond is 459 kJ/mol.
4. 459 X 2 = 918 kJ/mol.
5. This is the average potential energy that is present in 1 molecule of water. (This exercise can be used for calculating chemical bond energies in other molecules as well.)
So why is it important to know about bond energies?
Because, the amount of energy that is present in a bond energy is equivalent to the amount of energy reauired to break that bond and release the potential energy. This concept is important to understant when discussing potential energy diagrams.
The following diagrams show two different reactions taking place;
1. Exothermic Reaction
2. Endothermic Reaction
The activiation energy in these diagrams is the difference between the potential energy level of the transitional state and the potential energy of the reacting molecules. You will see in the following two diagrams that an exothermic reaction requires a smaller activation energy than an endothermic reaction. This is the case because in an exothermic reaction there is a net amount of energy that is released and in an endothermic reaction there is a net amount of energy that is abosorbed.
What is ∆ H?
∆ H is a symbol that represents the heat or enthalpy of a reaction, and it is the overall change in energy that occurs in a chemical reaction.
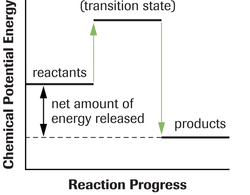
Exothermic Reaction
An exothermic reaction is one where;
∆ H = Negative Value
•There is an overall release of energy
An exothermic reaction is one where;
∆ H = Negative Value
•There is an overall release of energy
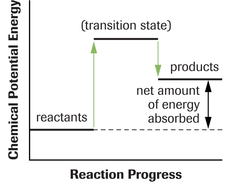
Endothermic Reaction
An endothermic reaction is one where;
∆ H = Negative Value
•There is an overall release of energy
So what does nature favour?
Exothermic Reactions!
Because....products of exothermic reactions have less potential energy and are therefore more stable.
DiGiuseppe, Maurice et al. (2003). Nelson Biology 12. Nelson Canada ELHI (1 Edition). Toronto, Ontario: Thomson Canada Limited
An endothermic reaction is one where;
∆ H = Negative Value
•There is an overall release of energy
So what does nature favour?
Exothermic Reactions!
Because....products of exothermic reactions have less potential energy and are therefore more stable.
DiGiuseppe, Maurice et al. (2003). Nelson Biology 12. Nelson Canada ELHI (1 Edition). Toronto, Ontario: Thomson Canada Limited
