INTERMOLECULAR FORCES
Now that we know how atoms interact with each other within a molecule, how do molecules interact with other molecules? The following intermolecular forces, also known as Van der Waals forces, are listed in increasing strength
Now that we know how atoms interact with each other within a molecule, how do molecules interact with other molecules? The following intermolecular forces, also known as Van der Waals forces, are listed in increasing strength
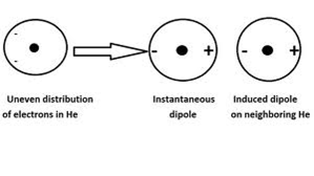
LONDON DISPERSION FORCES
London dispersion forces occur between all atoms and molecules. They are the only forces that are present between non-polar molecules and noble gases. Since electrons are constantly moving around the nucleus of an atom, at any given time, the electrons may be temporarily concentrated on one side of the molecule. This leads to a small, temporary negative charge on that side and a small, temporary positive charge on the other side.
If the slightly positive side comes close to a neighbouring molecule, it temporarily attracts the electrons in a neighbouring molecule creating a temporary induced dipole.
(Image retrieved from: http://chemwiki.ucdavis.edu/Physical_Chemistry/Quantum_Mechanics/Atomic_Theory/Intermolecular_Forces/London_Dispersion_Interactions
London dispersion forces occur between all atoms and molecules. They are the only forces that are present between non-polar molecules and noble gases. Since electrons are constantly moving around the nucleus of an atom, at any given time, the electrons may be temporarily concentrated on one side of the molecule. This leads to a small, temporary negative charge on that side and a small, temporary positive charge on the other side.
If the slightly positive side comes close to a neighbouring molecule, it temporarily attracts the electrons in a neighbouring molecule creating a temporary induced dipole.
(Image retrieved from: http://chemwiki.ucdavis.edu/Physical_Chemistry/Quantum_Mechanics/Atomic_Theory/Intermolecular_Forces/London_Dispersion_Interactions
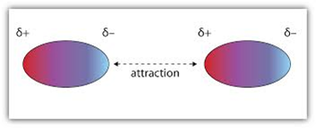
DIPOLE-DIPOLE INTERACTIONS
Dipole-dipole interactions hold polar molecules together. The slightly positive regions on one molecule are attracted to the slightly negative regions of another molecule
(Image retrieved from: http://catalog.flatworldknowledge.com/bookhub/reader/2273?e=ball-ch10_s01)
Dipole-dipole interactions hold polar molecules together. The slightly positive regions on one molecule are attracted to the slightly negative regions of another molecule
(Image retrieved from: http://catalog.flatworldknowledge.com/bookhub/reader/2273?e=ball-ch10_s01)
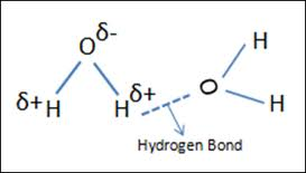
HYDROGEN BONDING (H-bonding)
Hydrogen bonds are especially strong dipole-dipole interactions. The electropositive hydrogen on one polar molecule is attracted to the electronegative N,O, or F on a neighbouring atom. H-bonding is demonstrated in water molecules. H-bonds occur between a hydrogen on one H2O molecule and the oxygen on a neighbouring H2O molecule. These strong intermolecular interactions lead to the unique properties of water: capillary action, temperature moderation, surface tension, evaporative cooling, and ice floating on water. (Image retrieved from: http://chemwiki.ucdavis.edu/Physical_Chemistry/Spectroscopy/Vibrational_Spectroscopy/Infrared_Spectroscopy/Infrared%3A_Interpretation)
Students may be curious to know why ice floats on water or why the polar ice caps float; if the molecules of water are colder, they should have less kinetic energy and would be moving more slowly. This would mean the molecules would be closer together (more dense). Since substances that are more dense sink and substances that are less dense float, shouldn't ice actually sink in water? The video below details the H-bonding that occurs in water and the reason why ice floats. (Video retrieved from: http://www.youtube.com/watch?v=UukRgqzk-KE)
Hydrogen bonds are especially strong dipole-dipole interactions. The electropositive hydrogen on one polar molecule is attracted to the electronegative N,O, or F on a neighbouring atom. H-bonding is demonstrated in water molecules. H-bonds occur between a hydrogen on one H2O molecule and the oxygen on a neighbouring H2O molecule. These strong intermolecular interactions lead to the unique properties of water: capillary action, temperature moderation, surface tension, evaporative cooling, and ice floating on water. (Image retrieved from: http://chemwiki.ucdavis.edu/Physical_Chemistry/Spectroscopy/Vibrational_Spectroscopy/Infrared_Spectroscopy/Infrared%3A_Interpretation)
Students may be curious to know why ice floats on water or why the polar ice caps float; if the molecules of water are colder, they should have less kinetic energy and would be moving more slowly. This would mean the molecules would be closer together (more dense). Since substances that are more dense sink and substances that are less dense float, shouldn't ice actually sink in water? The video below details the H-bonding that occurs in water and the reason why ice floats. (Video retrieved from: http://www.youtube.com/watch?v=UukRgqzk-KE)
