Photosynthesis
below you will find some common misconceptions regarding photosynthesis that students and teachers encounter
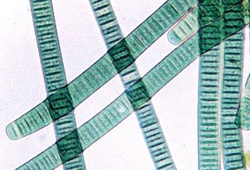
MISCONCEPTION #1: Cyanobacteria are sometimes called blue-green algae! The name “cyanobacteria” in fact comes from the Greek translation of the bacteria’s colour. It is incorrect to call them algae because cyanobacteria are prokaryotic, and algae are in fact eukaryotic.
Photo credit:
http://www.ucmp.berkeley.edu/bacteria/cyanointro.html
Photo credit:
http://www.ucmp.berkeley.edu/bacteria/cyanointro.html
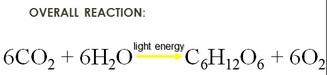
MISCONCEPTION #2: Students sometimes ignore the word “overall” and assume this is a one step reaction! As teachers, we have to stress the fact that not all reactants are used at once, and not all products are given off at once. The overall equation gives no indication of the complex process in which the overall change is accomplished.
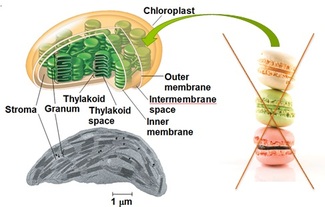
MISCONCEPTION #3: Thylakoids are similar to stacked cookies! In fact, the lumen (empty space) of all the stacked thylakoids (grana) and the unstacked thylakoids (lamellae) is connected!

MISCONCEPTION #4: Leaves that are not green are dying! In the picture on the right, we see a mosaic forest made up of different coloured trees within the same season (ex. Maple). Their accessory pigments such as carotene are arranged towards the top layer of the leaves, as an evolutionary advantage to attract pollinators. In the bottom picture, we are shown leaves in the summer and then in the fall. Leaves contain chlorophyll as well as other accessory pigments. In the summer, the green colour reflected by chlorophyll overwhelms the colour reflected by the other pigments, and we perceive trees as green. In the fall, leaves break down cholorphyll and conserve it for the spring.
Photo credit:
http://www.panoramio.com/photo/8127357
Photo credit:
http://www.panoramio.com/photo/8127357
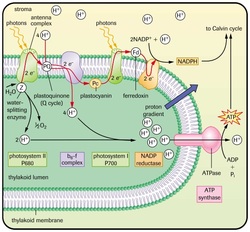
MISCONCEPTION #5: The blue arrow in this commonly used diagram is misleading! Protons can not get through the phospholipid bilayer. H+ accumulates in the lumen which makes the pH lower (more acidic). This means that electrochemical energy gets created inside.
MISCONCEPTION #6: ATP stores the most energy! In fact, NADPH stores way more energy than ATP because energy is stored in the H+. This misconception often arises from the fact that ATP is usually displayed in diagrams having an explosion start around it, whereas NADPH is displayed inside a simple rectangle.
MISCONCEPTION #6: ATP stores the most energy! In fact, NADPH stores way more energy than ATP because energy is stored in the H+. This misconception often arises from the fact that ATP is usually displayed in diagrams having an explosion start around it, whereas NADPH is displayed inside a simple rectangle.
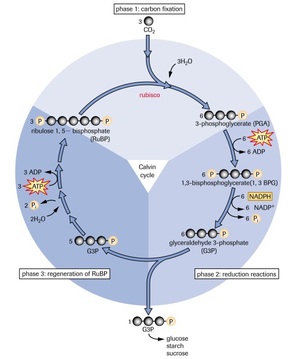
MISCONCEPTION #7: Calvin cycle can also be called the “dark reactions”. This is in fact incorrect. This process is still driven by light, but indirectly. This is because ATP and NADPH produced in the light-dependent reactions are key components which drive the Calvin Cycle. The correct terminology which could replace "the Calvin cycle" is "the light-independent reactions".
MISCONCEPTION #8: One Calvin cycle makes one glucose! In fact, 3 molecules of CO2 produce 6 molecules of G3P, one of which exits the cycle, so you need 6 molecules of CO2 in order to make one glucose which is formed by binding two G3P molecules. The Calvin cycle has to happen twice for one glucose molecule to be formed.
MISCONCEPTION #8: One Calvin cycle makes one glucose! In fact, 3 molecules of CO2 produce 6 molecules of G3P, one of which exits the cycle, so you need 6 molecules of CO2 in order to make one glucose which is formed by binding two G3P molecules. The Calvin cycle has to happen twice for one glucose molecule to be formed.
For additional misconceptions not mentioned here, visit the link below.

