BIOMOLECULES
The last section in the basic biochemistry topic is about biomolecules. These include carbohydrates, lipids, proteins, and nucleic acids. As nucleic acids are also covered in the DNA:Structure and Function section, the biomolecules covered in this section will be the first three only.The YouTube clip below could be used as an introduction or a general summary post-lesson. "Molecules Gone Wild" is a song to the tune of "Gangnam Style - Psy" about biomolecules, their structures and their functions.
The last section in the basic biochemistry topic is about biomolecules. These include carbohydrates, lipids, proteins, and nucleic acids. As nucleic acids are also covered in the DNA:Structure and Function section, the biomolecules covered in this section will be the first three only.The YouTube clip below could be used as an introduction or a general summary post-lesson. "Molecules Gone Wild" is a song to the tune of "Gangnam Style - Psy" about biomolecules, their structures and their functions.
Retrieved from: http://www.youtube.com/watch?v=nt9u7CfVoc4
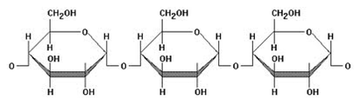
CARBOHYDRATES (CH2O)n
Carbohydrates are the most common organic materials on Earth. Their functions are storage of energy, structural support, and cell surface markers.
Monosaccharides (ie. glucose) are carbohydrate monomers - single chains of carbons with hydrogen atoms and hydroxyl groups attached. Oligosaccharides (ie. sucrose - glucose & fructose) are 2-3 monosaccharides attached via ether bonds (glycosidic linkages). An example of an oligosaccharide can be seen in the image above. Polysaccharides (ie. chitin) are hundreds to thousands of monosaccharides long and are also attached via glycosidic linkages.
(Image retrieved from: http://www.mansfield.ohio-state.edu/~sabedon/biol1025.htm)
Carbohydrates are the most common organic materials on Earth. Their functions are storage of energy, structural support, and cell surface markers.
Monosaccharides (ie. glucose) are carbohydrate monomers - single chains of carbons with hydrogen atoms and hydroxyl groups attached. Oligosaccharides (ie. sucrose - glucose & fructose) are 2-3 monosaccharides attached via ether bonds (glycosidic linkages). An example of an oligosaccharide can be seen in the image above. Polysaccharides (ie. chitin) are hundreds to thousands of monosaccharides long and are also attached via glycosidic linkages.
(Image retrieved from: http://www.mansfield.ohio-state.edu/~sabedon/biol1025.htm)
LIPIDS
Lipids are subdivided into four categories: fats, phospholipids, steroids, and waxes. Fats and phospholipids are the focus of this section and are described in detail below.
Lipids are subdivided into four categories: fats, phospholipids, steroids, and waxes. Fats and phospholipids are the focus of this section and are described in detail below.
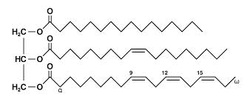
Fats
Fats are the most common energy storing molecules in living organisms - they store more than 2x the energy of carbohydrates and proteins. Fats occur in the form of triglycerides (image to the right), which are composed of a glycerol backbone and three fatty acids (hydrocarbon chains) linked by ester bonds.
Students may be interested to know that saturated fats refer to the saturation of the fatty acids with H atoms. If a fatty acid is saturated, it contains no double bonds. If the fat is monounsaturated, it contains a single double bond. Polyunsaturated fats contain multiple double bonds.
(Image retrieved from: http://commons.wikimedia.org/wiki/File:Fat_triglyceride_shorthand_formula.PNG)
Fats are the most common energy storing molecules in living organisms - they store more than 2x the energy of carbohydrates and proteins. Fats occur in the form of triglycerides (image to the right), which are composed of a glycerol backbone and three fatty acids (hydrocarbon chains) linked by ester bonds.
Students may be interested to know that saturated fats refer to the saturation of the fatty acids with H atoms. If a fatty acid is saturated, it contains no double bonds. If the fat is monounsaturated, it contains a single double bond. Polyunsaturated fats contain multiple double bonds.
(Image retrieved from: http://commons.wikimedia.org/wiki/File:Fat_triglyceride_shorthand_formula.PNG)
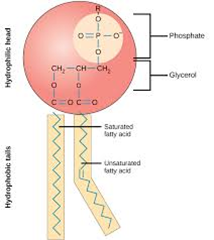
Phospholipids
Phospholipids have a phosphate glycerol head and a tail composed of two fatty acids.These components are linked via phosphate ester and ester bonds. Phospholipids are the main components of the lipid bilayer that exists in cell membranes. These phospholipids give cell membranes their fluid qualities. In colder weather, it is possible for these cell membranes to become rigid and freeze, which would would pose a problem for the cell. In order to prevent this, cell membrane phospholipids alter the bonding of the attached fatty acids Adding double bonds to the fatty acid chain, introduces a kink in the chain. The more double bonds that are present, the more kinks there are in the chain. If these chains are bent, they cannot stack as closely together as they could if they were straight. As such, bent chains lead to more fluid substances (oil or a more fluid membrane) and straight chains lead to more solid substances (butter or a less solid membrane).
In summary, warm weather creates a need for cell membranes to be more rigid. To attain this, cell membranes remove double bonds present in their phospholipid fatty acids. In colder weather, to become more fluid, cell membranes need to add double bonds to their phospholipid fatty acids.
(Image retrieved from: https://www.boundless.com/biology/biological-macromolecules/lipids/phospholipids/)
Phospholipids have a phosphate glycerol head and a tail composed of two fatty acids.These components are linked via phosphate ester and ester bonds. Phospholipids are the main components of the lipid bilayer that exists in cell membranes. These phospholipids give cell membranes their fluid qualities. In colder weather, it is possible for these cell membranes to become rigid and freeze, which would would pose a problem for the cell. In order to prevent this, cell membrane phospholipids alter the bonding of the attached fatty acids Adding double bonds to the fatty acid chain, introduces a kink in the chain. The more double bonds that are present, the more kinks there are in the chain. If these chains are bent, they cannot stack as closely together as they could if they were straight. As such, bent chains lead to more fluid substances (oil or a more fluid membrane) and straight chains lead to more solid substances (butter or a less solid membrane).
In summary, warm weather creates a need for cell membranes to be more rigid. To attain this, cell membranes remove double bonds present in their phospholipid fatty acids. In colder weather, to become more fluid, cell membranes need to add double bonds to their phospholipid fatty acids.
(Image retrieved from: https://www.boundless.com/biology/biological-macromolecules/lipids/phospholipids/)
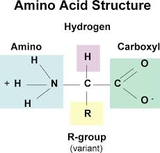
PROTEINS
Proteins are composed of amino acids. The general structure of amino acids are indicated in the image to the right. The properties of amino acids come from the interactions of the R groups (functional groups) that are attached to the general structure. Since there are 20 different R groups, there are 20 different possible amino acids. These are either polar, non-polar, or charged (acidic or basic) depending on the properties of the R groups. (Image retrieved from: http://www.proprofs.com/flashcards/story.php?title=biological-molecules-aqa-proteins)
The building of polypeptides involves condensation reactions to attach the amino group from one amino acid to the carboxyl group on the next amino acid. The resulting bond is called a peptide bond (amide linkage)
There are four levels of basic protein structure:
Proteins are composed of amino acids. The general structure of amino acids are indicated in the image to the right. The properties of amino acids come from the interactions of the R groups (functional groups) that are attached to the general structure. Since there are 20 different R groups, there are 20 different possible amino acids. These are either polar, non-polar, or charged (acidic or basic) depending on the properties of the R groups. (Image retrieved from: http://www.proprofs.com/flashcards/story.php?title=biological-molecules-aqa-proteins)
The building of polypeptides involves condensation reactions to attach the amino group from one amino acid to the carboxyl group on the next amino acid. The resulting bond is called a peptide bond (amide linkage)
There are four levels of basic protein structure:
- Primary: sequence of amino acids
- Secondary: hydrogen bonds between amino acids make α-helices and β-pleated sheets
- Tertiary: further folds of polypeptides stabilized by functional group interactions
- Quaternary: clustering of 2+ polypeptides in a tertiary structure
