ATOMIC STRUCTURE
Elements in the periodic table are arranged based on ascending atomic number. When looking at the table, the atomic number can be seen on the top of the element symbol and the electronegativity of the element is displayed underneath (the left image in the diagram below represents an element in the periodic table). Commonly, when referring to isotopes, the atomic number is listed on the bottom left of the element symbol and the mass number is listed on the upper left. Mass number represents the number of protons and neutrons, however the number of protons is remains constant. Isotopes are written in this fashion so that the number of neutrons (subtraction of protons from the mass number) is easy to determine. (Image below retrieved from: http://education-portal.com/academy/lesson/types-of-radioactive-decay-and-their-effect-on-the-nucleus.html#lesson)
Elements in the periodic table are arranged based on ascending atomic number. When looking at the table, the atomic number can be seen on the top of the element symbol and the electronegativity of the element is displayed underneath (the left image in the diagram below represents an element in the periodic table). Commonly, when referring to isotopes, the atomic number is listed on the bottom left of the element symbol and the mass number is listed on the upper left. Mass number represents the number of protons and neutrons, however the number of protons is remains constant. Isotopes are written in this fashion so that the number of neutrons (subtraction of protons from the mass number) is easy to determine. (Image below retrieved from: http://education-portal.com/academy/lesson/types-of-radioactive-decay-and-their-effect-on-the-nucleus.html#lesson)
Protons (positively charged) and neutrons (neutrally charged) are both located within the nucleus of the atom. Electrons (negatively charged) move around the nucleus in orbitals or energy shells. Atoms with equal numbers of protons and neutrons are neutrally charged. If there are more of one than the other, the atom is an ion and carries a charge. If there are more protons, the atom is a positive ion and if there are more electrons, the atom is a negative ion.The Bohr Rutherford model of the atom
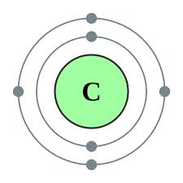
The Bohr Rutherford model of the atom is depicted to the left. Carbon has an atomic number of 6 which means it has 6 protons. For the atom to be neutrally charged, it also needs 6 electrons. Bohr Rutherford models depict the movement of electrons around the nucleus in orbitals. The first orbital can hold 2 electrons and subsequent orbitals can hold 8 (octet) that sit on the orbitals in pairs. When creating Bohr Rutherford diagrams, orbitals are not added until the previous orbital is full. Atoms are most stable and nonreactive when their orbitals have complete octets (or a full pair in the case of Helium - remember the first shell only holds 2 electrons!)
(Image retrieved from: http://sco.wikipedia.org/wiki/Carbon) |
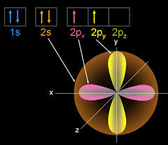
Orbital diagrams revolve around the random movement of electrons and depict electrons as clouds in energy levels. Energy level 1 is the same as the first orbital in Bohr Rutherford models and therefore can only hold 2 electrons. Energy level 2 can hold 8, etc. The diagram to the left depicts an orbital diagram of Carbon. The electrons are indicated by arrows and are still listed in pairs. (Image retrieved from: http://www.chemistryland.com/CHM151S/09-CovalentBonds/Covalent.html)
|
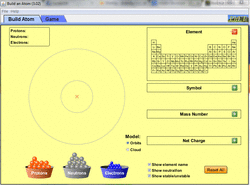
PhET Simulations are free simulations from the University of Colorado Boulder - Build an Atom is a great simulation for students to try at this point. It allows you to experiment on your own adding and subtracting protons, neutrons, and electrons while indicating which element, mass number, and charge you are currently depicting. There is also a game that varies in difficulty and allows students to build atoms according to different conditions or state the atom and conditions depicted. Clicking on the image to the left will bring you to the simulation page. (Simulation retrieved from: http://phet.colorado.edu/en/simulation/build-an-atom)

