FUNCTIONAL GROUPS
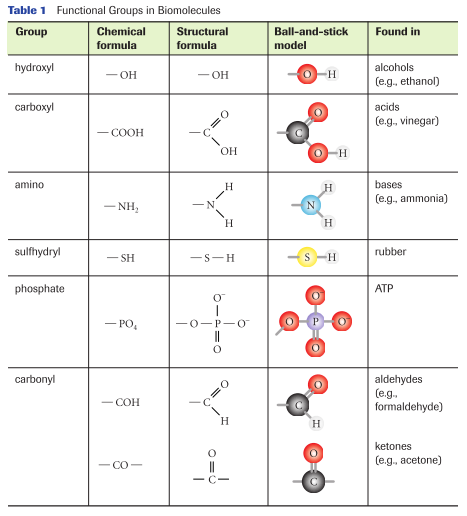
In the Atomic Structure section, we notes that atoms containing complete octets in their valence shell are nonreactive and stable. It is also safe to assume that atoms containing incomplete octets, are reactive. This makes sense when we remember that atoms want to maintain a complete octet to become stable. To do this, they need to steal electrons, share them, or have them taken away by another incomplete atom. In the same way, molecules will react with other molecules depending on their polarity. Biological molecules are largely made of hydrocarbons; a straight chain of hydrogen and carbon, however, is non-polar. Additional functional groups (reactive clusters of atoms attached to the backbone of an organic molecule) can give the hydrocarbon chain polarity; however, different functional groups have different properties. Below, you will find a chart listing names, structures, locations, and models of common functional groups found in biological molecules. As mentioned before, different atoms have different numbers of electrons in their valence shell; each missing electron represents one possible bond. For example, carbon has 4 valence electrons; to complete its octet, it requires four additional electrons. As such, carbon can make four possible bonds with other atoms. (Image retrieved from: Nelson Biology 12)
In the Atomic Structure section, we notes that atoms containing complete octets in their valence shell are nonreactive and stable. It is also safe to assume that atoms containing incomplete octets, are reactive. This makes sense when we remember that atoms want to maintain a complete octet to become stable. To do this, they need to steal electrons, share them, or have them taken away by another incomplete atom. In the same way, molecules will react with other molecules depending on their polarity. Biological molecules are largely made of hydrocarbons; a straight chain of hydrogen and carbon, however, is non-polar. Additional functional groups (reactive clusters of atoms attached to the backbone of an organic molecule) can give the hydrocarbon chain polarity; however, different functional groups have different properties. Below, you will find a chart listing names, structures, locations, and models of common functional groups found in biological molecules. As mentioned before, different atoms have different numbers of electrons in their valence shell; each missing electron represents one possible bond. For example, carbon has 4 valence electrons; to complete its octet, it requires four additional electrons. As such, carbon can make four possible bonds with other atoms. (Image retrieved from: Nelson Biology 12)
Give a Hair a Perm Lab
An interesting experiment demonstrating the properties of functional groups is provided below. This hair lab uses the bonding between neighbouring sulfhydryl groups (disulfide bond) to create permanently curly hair from a straight piece of hair. Because hair is made of keratin (a protein) and proteins contain amino acids (which also have functional groups), the folding of these proteins leads to sulfydryl functional groups coming into close contact. If this is the case, the sulfydryl groups form a disulfide bond, which creates a kink in the keratin. The more disulfide bonds present, the curlier the hair. (Lab retrieved from: http://ed.fnal.gov/arise/guides/bio/2-Biochemistry/2c-BiochemHairChemistryII.pdf)
An interesting experiment demonstrating the properties of functional groups is provided below. This hair lab uses the bonding between neighbouring sulfhydryl groups (disulfide bond) to create permanently curly hair from a straight piece of hair. Because hair is made of keratin (a protein) and proteins contain amino acids (which also have functional groups), the folding of these proteins leads to sulfydryl functional groups coming into close contact. If this is the case, the sulfydryl groups form a disulfide bond, which creates a kink in the keratin. The more disulfide bonds present, the curlier the hair. (Lab retrieved from: http://ed.fnal.gov/arise/guides/bio/2-Biochemistry/2c-BiochemHairChemistryII.pdf)
| 2c-biochemhairchemistryii.pdf | |
| File Size: | 84 kb |
| File Type: | |