CHEMICAL REACTIONS
There are four major chemical reactions that are detailed in the basic biochemistry section: neutralization, condensation, hydrolysis, and redox reactions.
There are four major chemical reactions that are detailed in the basic biochemistry section: neutralization, condensation, hydrolysis, and redox reactions.
NEUTRALIZATION REACTIONS
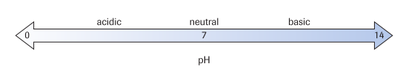
To understand neutralization reactions, we need to understand the autoionization of water to provide acids and bases. A very small proportion of water molecules transfer an H+ ion to another H2O leading to the formation of hydroxide ions (OH-) and hydronium ions (H3O+): H2O + H2O → H3O+ + OH-
[H3O+ ] = [OH-] = 10-7 mol/L; pH = 7 **In water, the concentration of hydronium ions is always equivalent to the concentration of hydroxide ions. because of this, the pH of water is 7 (neutral)
Acids: substances that increase [H3O+] when dissolved in water
HCl + H2O → H3O+ + Cl- pH < 7
Bases: substances that that increase [OH-] when dissolved in water. They may do this by:
(a) producing OH- directly (e.g., NaOH)
NaOH → Na+ + OH- pH > 7
(b) combine with H+ of a water molecule to make OH- (e.g., NH3)
NH3 + H2O → NH4+ + OH- pH > 7
Acids: substances that increase [H3O+] when dissolved in water
HCl + H2O → H3O+ + Cl- pH < 7
Bases: substances that that increase [OH-] when dissolved in water. They may do this by:
(a) producing OH- directly (e.g., NaOH)
NaOH → Na+ + OH- pH > 7
(b) combine with H+ of a water molecule to make OH- (e.g., NH3)
NH3 + H2O → NH4+ + OH- pH > 7
A neutralization occurs when an acid and a base react to form water and a salt. For example:
HCl+NaOH → H2O+NaCl
(Image above retrieved from Nelson Biology 12)
HCl+NaOH → H2O+NaCl
(Image above retrieved from Nelson Biology 12)
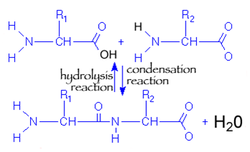
CONDENSATION AND HYDROLYSIS REACTIONS
Condensation and hydrolysis are opposite reactions. Condensation is anabolic; it builds polymers from monomers by releasing water. Hydrolysis, on the other hand, is catabolic; it breaks polymers into monomers and requires the use of water. The diagram to the right illustrates how these reactions are reverse processes.
(Image retrieved from:
http://virtuallaboratory.colorado.edu/Biofundamentals/lectureNotes/Topic3-4_Peptide%20bonds.htm)
Condensation and hydrolysis are opposite reactions. Condensation is anabolic; it builds polymers from monomers by releasing water. Hydrolysis, on the other hand, is catabolic; it breaks polymers into monomers and requires the use of water. The diagram to the right illustrates how these reactions are reverse processes.
(Image retrieved from:
http://virtuallaboratory.colorado.edu/Biofundamentals/lectureNotes/Topic3-4_Peptide%20bonds.htm)
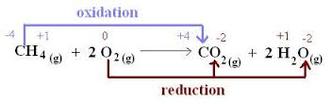
REDOX REACTIONS
Redox reactions (also known as oxidation-reduction reactions) are coupled reactions and are extremely important in understanding how and why the metabolic processes involved in the metabolism section of the SBI4U curriculum occur. Reduction and oxidation refer to the reduction and oxidation of electrons. This may be confusing for some students to understand and to help, you can remember the phrase "LEO the lion says GER", which refers to loss of electrons is oxidation and gain of electrons is reduction. They can also remember "OIL RIG", which represents oxidation is loss and reduction is gain. Most energy transfer in cells is via oxidation-reduction reactions and the oxidation of one molecule means the reduction of another. Redox reactions may occur in one of three ways:
(Image retrieved from: http://chemistry.tutorvista.com/inorganic-chemistry/redox-potential.html)
Redox reactions (also known as oxidation-reduction reactions) are coupled reactions and are extremely important in understanding how and why the metabolic processes involved in the metabolism section of the SBI4U curriculum occur. Reduction and oxidation refer to the reduction and oxidation of electrons. This may be confusing for some students to understand and to help, you can remember the phrase "LEO the lion says GER", which refers to loss of electrons is oxidation and gain of electrons is reduction. They can also remember "OIL RIG", which represents oxidation is loss and reduction is gain. Most energy transfer in cells is via oxidation-reduction reactions and the oxidation of one molecule means the reduction of another. Redox reactions may occur in one of three ways:
- By transfer of electron(s) from one atom to another
- By transfer of H atom(s) from one compound to another (H atoms carry electrons)
- By transfer of O atom(s) from one compound to another (O atoms draw electrons away from compounds because it is highly electronegative)
(Image retrieved from: http://chemistry.tutorvista.com/inorganic-chemistry/redox-potential.html)