electron Transport and Chemiosmosis
NADH and FADH2 transfer the hydrogen atom electrons to a series of proteins in the inner mitochondrial membrane called the electron transport chain.
- The first protein complex (NADH dehydrogenase) picks up the hydrogen atom (with 2 electrons) from NADH and ubiquinone (Q), an electron carrier, strips the electrons which causes the protein complex to let go of the proton and the proton translocates into the intermembrane space.
- Q shuttles the electrons to the next complex (cytochrome b-c1 complex). The first protein complex is oxidized and the second is reduced.
- As the electrons move between molecules they occupy more and more stable positions relative to nuclei. The free energy released is used to move H+ ions (protons) across the inner mitochondrial membrane.
- Cytochrome C will shuttle 2 electrons to the next protein complex (cytochrome oxidase complex). Once again free energy is released by electrons becoming more stable and it is used to translocate H+ across membrane.
- Finally, the cytochrome oxidase enzyme catalyzes the reaction between the electrons, protons, and oxygen to form water. At this point we need an extremely electronegative substance to oxidize the last protein because the electrons at this point are extremely stable. Oxygen can do this and with 2 H+ from the matrix water is formed. Without oxygen this process would back up and not be able to occur.
- The process is highly exergonic and the free energy is used to translocate protons across the membrane and set up a proton gradient across the inner mitochondrial membrane.
NADH vs. FADH2
- NADH can pass its electrons to the first protein complex in the electron transport chain.
- FADH2 transfers its electrons first to Q (ubiquinone).
- NADH can pump 3 protons.
- FADH2 can only pump 2 protons.
- As a result, NADH forms 3 ATP molecules and FADH2 forms 2 ATP molecules.
- NADH produced by glycolysis in the cytoplasm is brought into the matrix by a glycerol-phosphate shuttle and converts it to FADH2.
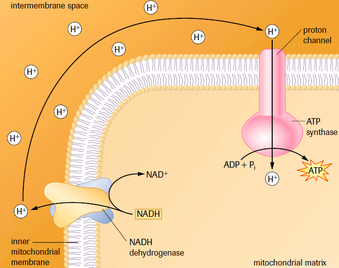
Chemiosmosis and Oxidative ATP Synthesis
- Remember: there is now an electrochemical gradient that is storing free energy.
- Electrical component: higher positive charge in the intermembrane space than the matrix.
- Chemical component: higher concentration of protons in the intermembrane space than the matrix.
- This gradient creates a voltage across the membrane much like a battery.
- The protons are unable to pass through the phospholipid bilayer unaided.
- The protons cannot diffuse through the phospholipid bilayer alone. They are forced through special proton channels associated with ATPase.
- The free energy moves protons through the ATPase.
- This energy drives the synthesis of ATP from ADP and Pi in the matrix.
- Peter Mitchell received the Nobel Prize in chemistry in 1978 for discovering this mechanism of ATP generation. He called the process chemiosmosis because the energy that creates the ATP comes from osmosis of protons through a membrane.
Student Activity: Model Making & Videos
What Next?
ATP molecules are transported through the mitochondrial membranes by facilitated diffusion into the cytoplasm of the cell where they can drive endergonic processes like movement, active transport, and synthesis reactions in the cell.
Energy Efficiency
ATP molecules are transported through the mitochondrial membranes by facilitated diffusion into the cytoplasm of the cell where they can drive endergonic processes like movement, active transport, and synthesis reactions in the cell.
Energy Efficiency
- By the end of aerobic cellular respiration, 36 ATP have been produced.
- The actual yield will be less than this because the inner mitochondrial membrane is not completely impermeable to H+ ions so some leak back without going through ATPase.
- Some protons in the reservoir are used by the cell for other energy-requiring activities.
- The actual yield is closer to 30 ATP per molecule of glucose.
- Using the actual yield of 30 ATP per glucose molecule we can calculate the energy harvested from glucose:
- Efficiency = 30 mol ATP x 31 kJ/mol ATP / 2870 kJ x 100% = 32%
- This is much more efficient than glycolysis!
- In comparison, the energy efficiency of a car is approximately 25%.
| SmartBoard Activity: Energy Balance Worksheet | |
| File Size: | 339 kb |
| File Type: | notebook |