BONDING AND POLARITY
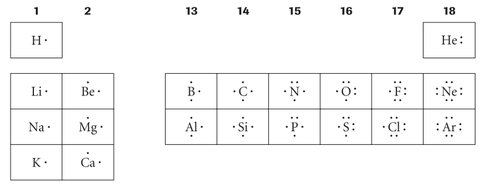
The outermost shell of an atom is called the valence shell. The valence shell becomes important when discussing bonding of atoms. As mentioned in atomic structure, atoms are nonreactive when they have full octets. Since you need to fill the innermost orbitals first, it can be assumed that a full octet in the valence shell means all inner orbitals are also full. Lewis Dot diagrams are drawn based on this assumption and are simple drawings that include only the number of electrons in their valence shells. Disregarding the transition metals in the periodic table, the number of valence electrons can be determined by the group (column) number. Since all atoms want to be stable, and to be stable they need to have full octets, elements want to gain the missing electrons and to do that, they need to bond with other atoms. This is also known as the octet rule.
As an example, the diagram below indicates that Carbon has four valence electrons. According to the octet rule, it wants to gain four more to have a full valence shell. To do this, it needs to bond with other atoms.
(Image retrieved from: Nelson Biology 12)
The outermost shell of an atom is called the valence shell. The valence shell becomes important when discussing bonding of atoms. As mentioned in atomic structure, atoms are nonreactive when they have full octets. Since you need to fill the innermost orbitals first, it can be assumed that a full octet in the valence shell means all inner orbitals are also full. Lewis Dot diagrams are drawn based on this assumption and are simple drawings that include only the number of electrons in their valence shells. Disregarding the transition metals in the periodic table, the number of valence electrons can be determined by the group (column) number. Since all atoms want to be stable, and to be stable they need to have full octets, elements want to gain the missing electrons and to do that, they need to bond with other atoms. This is also known as the octet rule.
As an example, the diagram below indicates that Carbon has four valence electrons. According to the octet rule, it wants to gain four more to have a full valence shell. To do this, it needs to bond with other atoms.
(Image retrieved from: Nelson Biology 12)
There are two types of atomic bonds - ionic and covalent.
IONIC COVALENT
IONIC COVALENT
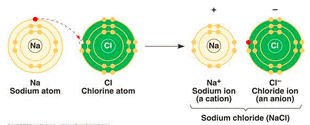
Ionic bonds are formed between metals and non-metals. In ionic bonds, one atom steals an electron from the other atom to complete their octet. Since electrons are negative, the atom that loses an electron becomes positively charged and the atom that takes the electron becomes negatively charged. From the atomic structure section, we know that charged atoms are called ions - positive ions are termed cations and negative ions are termed anions. (Image retrieved from: http://semoneapbiofinalexamreview.wikispaces.com/C.+Biochemistry+(2-3)) |

Covalent bonds are formed between two atoms that share an electron pair. Instead of one atom stealing an electron, the atoms share them. There are two types of covalent bonds, polar covalent bonds and non-polar covalent bonds. Non-polar covalent bonds occur when the two atoms share the electron pair equally. Polar covalent bonds occur when the two atoms share the electron pair unequally and one atom has a stronger pull. The atom with the stronger pull becomes slightly negative and the other becomes slightly positive. (Image retrieved from: http://en.wikipedia.org/wiki/Covalent_bond) (Note: this does not mean that the electron pair is permanently closer to one atom than the other. Remember that electrons are always moving. Polar covalent bonds only mean that the electron pair spends more of its time around one atom than the other) |

To the left is a visual representation of ionic, polar covalent, and non-polar covalent bonds in terms of relative pull (stronger pulls are represented by larger circles). (Image retrieved from: http://schools.birdville.k12.tx.us/cms/lib2/TX01000797/Centricity/Domain/912/ChemLessons/Lessons/Bonding/Chemical%20Bonding.htm)
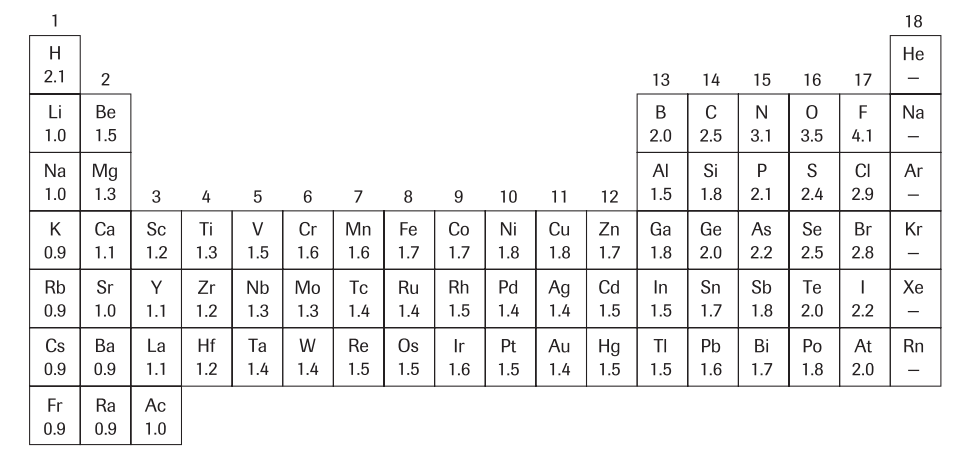
To determine whether a bond will be ionic, polar covalent, or non-polar covalent, we need to know a little bit about electronegativity or the strength of an atom's pull on an electron. A periodic table with the electronegativities is provided below. The type of bond is determined by the difference in electronegativity (ΔEn). (Image below retrieved from: Nelson Biology 12)
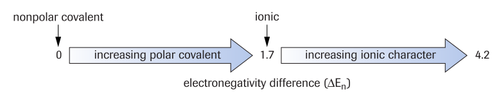
If the difference in electronegativities is above 1.7, the bond is ionic. This makes sense if you look back at the metals and non-metals in the periodic table. The metal elements have high electronegativities (strong pulls) and the non-metal elements have low electronegativities (weak pulls). Therefore, the difference in electronegativity is large. Since the metal atom has the larger electronegativity, it steals the electron from the non-metal and is subsequently the cation. The reverse is true for the non-metal and this is therefore the anion.
If the difference in electronegativities is 0, which is always the case when you have bonding between two of the same atoms, the bond is non-polar and there is equal sharing of the electron pair
If the difference in electronegativities is between 0 and 1.7, the bond is polar with the atom with the larger electronegativity being slightly negative and the atom with the lesser electronegativity being slightly positive.
These facts are summarized in the diagram to the right. (Image retrieved from: Nelson Biology 12)
If the difference in electronegativities is 0, which is always the case when you have bonding between two of the same atoms, the bond is non-polar and there is equal sharing of the electron pair
If the difference in electronegativities is between 0 and 1.7, the bond is polar with the atom with the larger electronegativity being slightly negative and the atom with the lesser electronegativity being slightly positive.
These facts are summarized in the diagram to the right. (Image retrieved from: Nelson Biology 12)
Below is a SMARTboard activity (created by myself) where students can rank bonds in the order of increasing difference in electronegativity. They are also able to place the boundaries between non-polar/polar covalent and polar covalent/ionic.
| electronegactivity.notebook | |
| File Size: | 12 kb |
| File Type: | notebook |