Entropy
- The measure of randomness (also known as disorder) in a collection of objects or energy
- Entropy increases when disorder increases
- Entropy is symbolized with the letter S
How can we explain entropy to students?
The topic of entropy can be a very tricky one to teach. It is important to use as many examples as possible when explaining this concept. One trick to use in the classroom is to let a deck of cards fall from their original pile. This notion depicts the increase in entropy because the cards are becoming more randomized in their position. A new package of cards is much more ordered than a pile of cards that has just fallen. The reason why a brand new card is more ordered is because energy had to go into putting the deck of cards in a specific order.
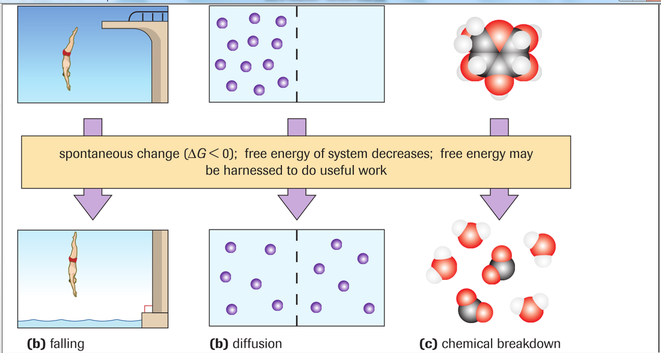
The diagram above shows the changes in entropy in three scenarios. In all three scenarios entropy is higher in the bottom illustration.
A) Falling: In this diagram it is important to note that in order for the diver to get to the platform he had to conduct work and use energy to get to the top. Thus, he ordered himself into that position. Him falling from that position is much more disordered because he simply fell and no energy was put into falling, and his fall is more randomized. This is why his fall is more disordered and thus more entropy is present as he his falling.
B) Diffusion: It would be more work to keep all the molecules on the left side of the membrane (as in the top illustration). Naturally, during diffusion molecules move from a higher concentration to a lower conentration. This is why there is more randomness and entropy in the bottom illustration.
C) Chemical Breakdown: It takes more energy to keep the molecules bound together, as well as to combine those molecules together to create such a compund. Thus if left to nature the compound would naturally decompose and the amount of entropy would increase.
So what does nature favour?
NATURE FAVOURS AN INCREASE IN ENTROPY!
WHY??
The second law of thermodynamics
What does the second law of thermodynamics state?
The entropy of the universe increases with any changes that occur. (Thus, why nature favours an increase in entropy).
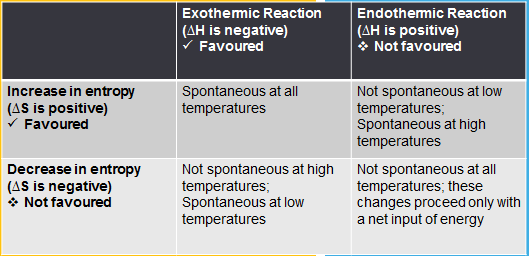
Sooo.... Now that we have learnt about ∆S and ∆H we can determine whether a
reaction will be spontaneous or not. The table below indicates how to determine what is happening.
A) Falling: In this diagram it is important to note that in order for the diver to get to the platform he had to conduct work and use energy to get to the top. Thus, he ordered himself into that position. Him falling from that position is much more disordered because he simply fell and no energy was put into falling, and his fall is more randomized. This is why his fall is more disordered and thus more entropy is present as he his falling.
B) Diffusion: It would be more work to keep all the molecules on the left side of the membrane (as in the top illustration). Naturally, during diffusion molecules move from a higher concentration to a lower conentration. This is why there is more randomness and entropy in the bottom illustration.
C) Chemical Breakdown: It takes more energy to keep the molecules bound together, as well as to combine those molecules together to create such a compund. Thus if left to nature the compound would naturally decompose and the amount of entropy would increase.
So what does nature favour?
NATURE FAVOURS AN INCREASE IN ENTROPY!
WHY??
The second law of thermodynamics
What does the second law of thermodynamics state?
The entropy of the universe increases with any changes that occur. (Thus, why nature favours an increase in entropy).
Sooo.... Now that we have learnt about ∆S and ∆H we can determine whether a
reaction will be spontaneous or not. The table below indicates how to determine what is happening.
DiGiuseppe, Maurice et al. (2003). Nelson Biology 12. Nelson Canada ELHI (1 Edition). Toronto, Ontario: Thomson Canada Limited