MOLECULAR POLARITY
Now we know how to determine if a bond is polar, but how do we know if an entire molecule is polar? We need to consider the geometry of the molecule of interest. VSEPR theory (Valence Shell Electron Pair Repulsion) suggests that electron pairs surrounding each atom and being negative, repel each other and move as far apart as possible. Below is an activity you can use in class to introduce the concept of molecular geometry.
Now we know how to determine if a bond is polar, but how do we know if an entire molecule is polar? We need to consider the geometry of the molecule of interest. VSEPR theory (Valence Shell Electron Pair Repulsion) suggests that electron pairs surrounding each atom and being negative, repel each other and move as far apart as possible. Below is an activity you can use in class to introduce the concept of molecular geometry.

Candy VSEPR Activity
Divide students into groups and give each group a different number of bonds, which are represented by toothpicks. Instruct them to build their molecule according to VSEPR theory (before you show them the diagrams). The peripheral atoms, which in this case are swedish berries (although any soft, light candy would work) must extend from the central atom, which in this case is a caramel. You can also have students measure the bond angles of their geometries to check for maximum repulsion. Following the activity, display the corresponding VSEPR diagrams to check for accuracy.
Divide students into groups and give each group a different number of bonds, which are represented by toothpicks. Instruct them to build their molecule according to VSEPR theory (before you show them the diagrams). The peripheral atoms, which in this case are swedish berries (although any soft, light candy would work) must extend from the central atom, which in this case is a caramel. You can also have students measure the bond angles of their geometries to check for maximum repulsion. Following the activity, display the corresponding VSEPR diagrams to check for accuracy.
- Remind students that molecules exist in 3D
- Variation: to add more complex geometries, give students larger candies (in the image above, these are represented by marshmallows) to represent non-bonding electrons. Instruct students that non-bonding electron pairs on the central atom take more space than bonding pairs.
- Variation: instead of using candy, you can also use balloons to represent electron clouds. The ties at the middle represent the central atom
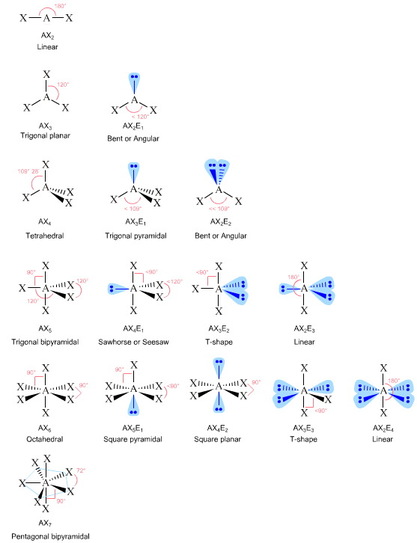
Geometries can also be polar or non-polar. Symmetrical geometries are non-polar because individual bond polarities cancel each other and asymmetrical geometries are potentially polar, however, you need to consider whether the individual bonds are polar. If the individual bonds are not polar, then the molecule cannot be polar. Below is a diagram of all VSEPR geometries with names and associated bond angles. (Image below retrieved from: http://jahschem.wikispaces.com/vsepr)
PhET simulations also has a free simulation for molecular polarity. It allows students to practice altering electronegativities of two or three atom molecules and indicate partial charges, bond polarities, and molecular polarities. It also lets students experiment with common biologically relevant molecules. Clicking the image to the left will bring you to the simulation.
(Simulation retrieved from: http://phet.colorado.edu/en/simulation/molecule-polarity)
(Simulation retrieved from: http://phet.colorado.edu/en/simulation/molecule-polarity)
WATER
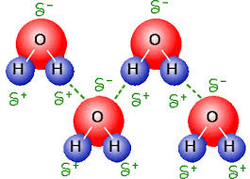
Water is called 'the universal solvent' because it dissolves more substances than any other liquid. This is due to its partial charges. Substances that are polar dissolve in water, and are said to be miscible. Substances that are non-polar do not dissolve in water, and are said to be immiscible. Just remember 'like dissolved like'. A biologically relevant example of this would be the use of hemoglobin for transport of CO2 and O2 in blood. Both O2 and CO2 are immiscible in water (and therefore blood). As such, they need to bind to hemoglobin, which IS miscible in water (and therefore blood) in order for their transport throughout the body.
(Image retrieved from: http://dgusvandi.blogspot.ca/2012/04/hydrogen-bonds.html)
Water is called 'the universal solvent' because it dissolves more substances than any other liquid. This is due to its partial charges. Substances that are polar dissolve in water, and are said to be miscible. Substances that are non-polar do not dissolve in water, and are said to be immiscible. Just remember 'like dissolved like'. A biologically relevant example of this would be the use of hemoglobin for transport of CO2 and O2 in blood. Both O2 and CO2 are immiscible in water (and therefore blood). As such, they need to bind to hemoglobin, which IS miscible in water (and therefore blood) in order for their transport throughout the body.
(Image retrieved from: http://dgusvandi.blogspot.ca/2012/04/hydrogen-bonds.html)